 :max_bytes(150000):strip_icc()/physical-and-chemical-changes-examples-608338_FINAL-f4e256e7fbf54f46a8c7bcefb300f5db.png)
So here, let's take a look at this example question. Now that we've seen this, let's click on the next video and take a look at an example question. Whenever dealing with chemical properties. Use this to guide you to the correct question, correct answer on a question. Remember when we're talking about chemical reactions, we're talking about chemical changes. So just remember connected to chemical properties is the idea of chemical reactions. But for now, just realize that saw the ability represents a chemical property. If the substance is not soluble, then it will not be thoroughly broken down by the liquid will go in greater detail in the idea of scalability in chapters later on. If the compound is soluble, it'll be broken down by the liquid. Ability now sell ability has to do with how our solid compound when it gets thrown into liquid, how the liquid and interact with one another. Now, finally, we have here this purple substance being dumped into a solvent, a liquid. So reactivity represents a chemical property. This is telling us that a reaction is a curry. Remember a chemical reaction sometimes causes a change that we can visually see that could be a color change or in this case the formation of bubbles. Next we have a beaker and we have bubbles emanating from it. Okay, so we'd say here that this is an image for corrosion. Is this substance toxic? Is it toxic to me? Can it slowly be hurting me? Next we have an image of liquid coming from little vials and when they drip, they break down whatever they're touching, that's because they're corrosive. But here when we use this symbol, it means toxicity. We know that this represents death in some way. So, basically, how easy is it for something to catch fire. 
So we're gonna say that a common type of chemical property is flammability. So if we take a look here for the first one, we have the image of a flame. Now with chemical properties, we have certain types of properties that are key to remember. So when we're talking about chemical properties, we must always keep in mind that it's related in some way to a chemical reaction where are reacting are starting material is changed chemically with its bonds and composition to help create something new. Remember we call these compounds after the arrows products. So two carbon dioxide gasses plus four water vapors here. So before the arrow, we say that these two compounds represent react ints they undergo a chemical change to produce C. The name of this compound would be called methanol. After the reaction here we have our chemical reaction, we have two CH three oh H. Now we're gonna say here the matter will be changed into a new substance. Remember a chemical reaction represents a chemical change where the composition and therefore the identity of a substance is forever altered. We're going to say that a chemical property is just a property of matter that is observed during a chemical reaction. Now here we're taking a look at chemical properties. We know that it can undergo physical and chemical changes with them being reversible or irreversible now associated with these changes? We can talk about the properties that accompany them. The differences between the two types of change are summarised below.So we've talked about matter and the different types of changes that it can undergo. These physical changes are often changes in state such as melting, freezing, boiling, condensing and dissolving. The other kind of change in chemistry is a physical change, which is when a substance changes without becoming a new chemical.  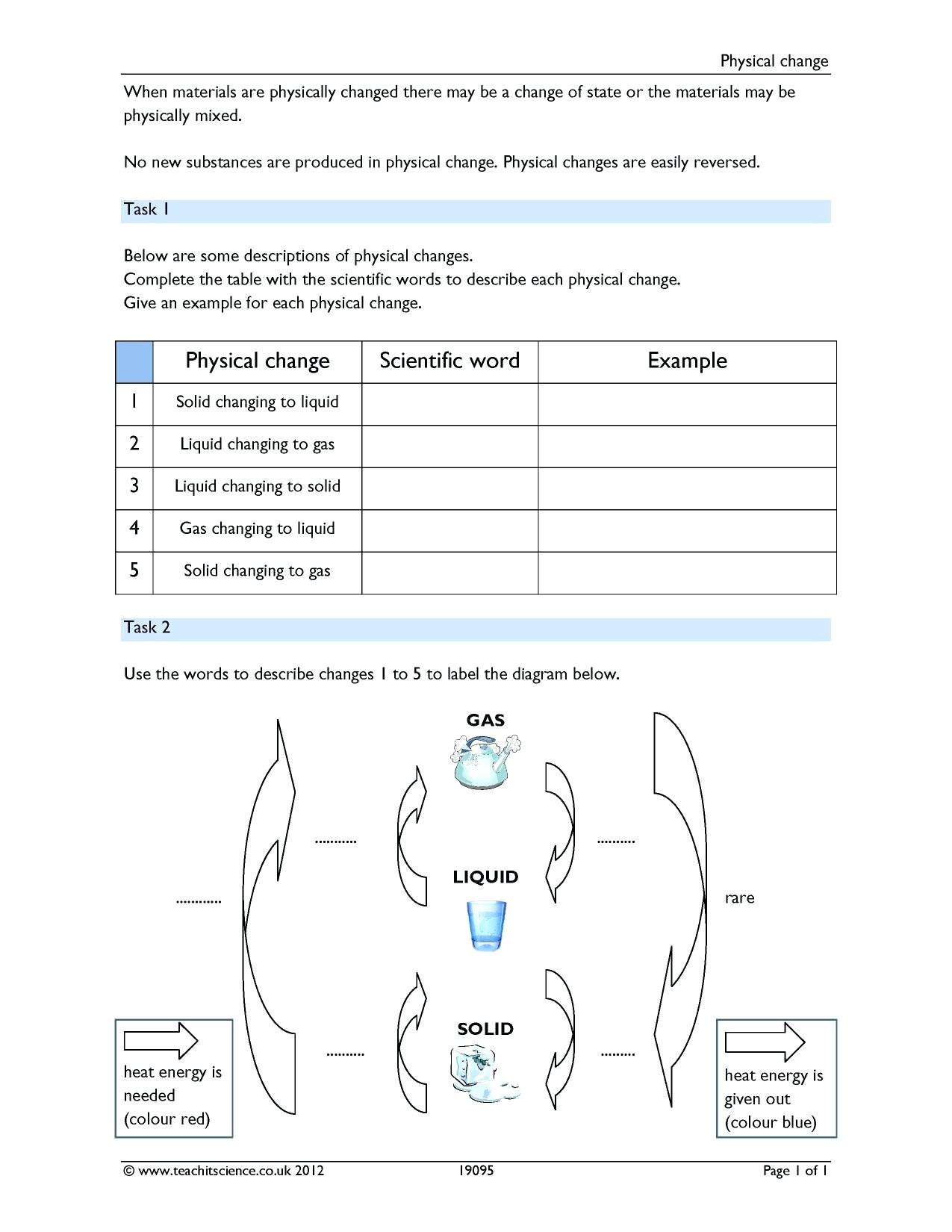
Instead, the atoms rearrange themselves to form new chemicals. When a chemical reaction occurs, atoms are neither created nor destroyed. Chemical and physical change Chemical change 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
